
In the second of two articles on gas planning for rebreather divers, BSAC Technical Group leader Mike Rowley takes a look at bailout strategies.
The greatest benefits of closed-circuit rebreather (CCR) diving are the huge advantage in gas efficiency plus the automatic mixing of gas to achieve the optimum gas mix at all stages of the dive.
A three-litre cylinder of oxygen plus a three-litre cylinder of diluent, for example, gives a theoretical 10 hours of gas duration and a CCR achieves this at all depths. In addition, the constant, optimum gas mix minimises decompression obligation as well as removing the problems associated with inappropriate gas at incorrect depths. But what if something goes wrong?
Bailing out onto open circuit is, for a CCR diver, a necessary emergency response to a rebreather problem. Such open-circuit bailout can be used in one of two ways:
- To provide a temporary safe gas to breathe and some thinking time, to find a solution to the problem and enable a return to closed-circuit breathing.
- As a final solution to a CCR failure that cannot be resolved and requires a safe alternative return to the surface, such as a complete loop flood or onset of carbon dioxide toxicity.
The moment a CCR diver bails out to open circuit they move from massive gas reserves and optimised variable gas mix to a limited gas supply and fixed gas mixes. The move to open circuit brings with it all of the issues and protocols of open circuit mixed-gas diving but, because this is an emergency response to a failure, the priorities for gas choice change significantly. The new priorities are to have:
- A safe gas to breathe at maximum operating depth (MOD).
- Safe gases to enable a return to the surface.
- Sufficient gas to return safely to surface.
 On the face of it, there isn’t a problem in terms of gas choice if we follow the open-circuit guidelines outlined in my In The Mix article on open-circuit mixed-gas planning in May’s SCUBA (issue #66). There are, however, subtle complexities involved that need to be considered. A rebreather dive is likely to involve less decompression obligation than a similar open-circuit dive if all goes according to plan.
On the face of it, there isn’t a problem in terms of gas choice if we follow the open-circuit guidelines outlined in my In The Mix article on open-circuit mixed-gas planning in May’s SCUBA (issue #66). There are, however, subtle complexities involved that need to be considered. A rebreather dive is likely to involve less decompression obligation than a similar open-circuit dive if all goes according to plan.
However, if an open-circuit bailout becomes necessary that decompression obligation can increase dramatically. There are additional issues of cold and the need to urinate involved in extended dive durations, but the immediate issue is gas provision. There are a number of ways we can deal with this. We can:
- Choose the bailout gases carefully to minimise the remaining duration of the dive.
- Adopt a gas-conserving strategy to maximise the gas supply.
- Adjust the parameters of the decompression plan to facilitate a reduced duration at the expense of conservatism.
Air diluent, nitrox bailout
Choosing the bailout gases carefully to minimise the remaining duration of the dive is relatively easy when using an air diluent: just choose the highest fraction of oxygen nitrox mix that is safe at the MOD. This will give the minimum decompression obligation in the event of an open-circuit bailout.
The question is what is the highest partial pressure of oxygen (PO2) that is safe at a given depth? The widely accepted maximum PO2, and the value BSAC recommends as a maximum is 1.4 bar. However open-circuit bailout is an emergency situation and as we want to minimise the decompression obligation to conserve gas, plus we will either be temporarily on the bailout gas while sorting the problem or when beginning an ascent, therefore, reducing the PO2 we may reasonably opt for choosing a gas with a PO2 of 1.6 bar at MOD.
For example, if we dive to a depth of 40 metres with an air diluent then the bailout gas using a PO2 at MOD of 1.6 bar
can be calculated using Dalton’s Law FO2 = PO2 / Pabs. Where FO2 = fraction of oxygen. Pabs = Absolute pressure.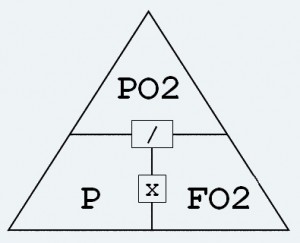 Or if you prefer to use Dalton’s triangle place your finger over the desired outcome (FO2) and the result will be PO2 divided by Pabs. For our 40m dive, the fraction of oxygen is 1.6 / 5 = 0.32.
Or if you prefer to use Dalton’s triangle place your finger over the desired outcome (FO2) and the result will be PO2 divided by Pabs. For our 40m dive, the fraction of oxygen is 1.6 / 5 = 0.32.
Therefore the open-circuit bailout gas for this dive would be nitrox 32 percent.
The strategy is to balance the risk of a short-term or quickly reducing PO2 of 1.6 bar and a similarly temporary high gas density of 6.2 grams/litre against the risk of running out of bailout gas and/or potentially inadequate decompression. To put it more bluntly we are accepting a temporary slightly higher risk of CNS toxicity and work of breathing to achieve a reduction in required gas.
Mixed gas, more complex bailout
When diving deeper with a mixed-gas diluent, we need to consider at least two different bailout gas mixes, one for
the deep part of the dive and another for the shallow. We have considered how to address the shallow portion of a
bailout ascent above, but we will need to determine a switch depth and therefore mix for the shallow gas.
First, let’s consider the choice of deep bailout gas mix. Currently, decompression models penalise divers for increased helium content in the breathing mix. It is often termed the helium penalty and tends to give deeper decompression stops and increased overall decompression obligation for the same depth and duration with increased helium. There is some interesting recent research that contradicts this assumption and may lead to different decompression models, but for the moment this is the case we have to contend with. In terms of gas density and narcosis more helium is good, but in terms of duration of bailout ascent, it will give us a longer duration with accompanying larger volume of gas required. It is a question of balancing the risks.
Keeping with the strategy of trying to balance risks against minimising duration and therefore the volume of bailout gas required, we need to consider our choice of gas.
Do we reduce the helium content by increasing the nitrogen content, thus managing the gas mix so that we accept more narcosis, isobaric counter diffusion (ICD) and greater gas density at the point of bailout in order to minimise the duration of the bailout ascent? It is worth remembering here that ICD becomes a risk when we switch to open circuit and a prudent strategy for avoiding ICD is to keep the PN2 differential at point of gas switch to about 0.5 bar or less.
Alternatively, do the dangers of greater gas density, ICD and more narcosis outweigh the requirement to carry more gas and spend more time in the water? Bearing in mind that a bailout that needs to go all the way to the surface is the result of some major failure with the rebreather then the diver will be stressed and experiencing massively increased breathing rates (RMV). RMVs above 80 litres a minute have been recorded in emergency situations. The Health and Safety Executive recommends planning for RMV of 50-75 litres per min until a point of safety is reached, usually the end of the first decompression stop.
What deep bailout gas?
Not only is the diver stressed, but so is their regulator. In these conditions, it is probably prudent to minimise the risk by using a higher helium content and accepting the requirement to carry a greater gas volume.
The recommended values of gas density of 5.2-6.2 grams/litre and narcosis at an END of 30-40m are probably a good guide to achieving this. So if we take a maximum PO2 of 1.6 bar and END of 25m (3.5 bar) we can calculate the optimum deep bailout gas.
Let us consider a dive to 60m, where Pabs is 7 bar. Using Dalton’s Law, a PO2 of 1.6 bar would give us an oxygen fraction of 1.6 / 7.0 = 0.229, that is 22 percent oxygen.
In Part 1 of this article in October’s SCUBA (issue #71), we saw how to calculate the END of a gas mix using FN2 = (END x 0.79) / Pabs.
On the 60m dive, FN2 will be (3.5 x 0.79) / 7 = 0.395, that is 40 per cent nitrogen.
Now that we know the oxygen and nitrogen content of the gas mix, it is just a matter of subtracting their sum from 100 percent to find the helium fraction. So, the percentage of helium in the mix will be 100 – (22 + 40) = 38, that is 38 percent helium.
We can now carry out a gas-density audit of the 22 / 38 mix, at MOD, using the known relative gas densities of oxygen, nitrogen and helium, to check it against our safe limits.
| Helium | 0.179 |
| Nitrogen | 1.251 |
| Oxygen | 1.428 |
For each gas we work out Fgas x gas density x Pabs.
For oxygen it’s 0.22 x 1.428 x 7 = 2.199
For nitrogen it’s 0.40 x 1.251 x 7 = 3.503
For helium it’s 0.38 x 0.179 x 7 = 0.476
Adding the three densities together gives a total gas density at MOD of 6.178 grams per litre.
It is worth noting that the gas density is just within the upper recommended limit while the END is well below the lower recommended limit. The gas density is the defining value.
Prudent to consider ICD
As I mentioned above, ICD is an issue when changing gases and a PN2 differential of 0.5 bar at the point of switching is a prudent avoidance strategy. Therefore it is wise to check that at the point of switching to bailout the PN2 of the bailout gas is not 0.5 bar greater than the PN2 of the loop gas.
From Dalton’s Law, or triangle if you prefer, the PN2 of the deep bailout gas at MOD is PN2 = Pabs x FN2.
In our example, the PN2 in our bailout gas trimix 22 / 38 at 60m will be 7 x 0.4 = 2.80 bar.
On our 60m dive, planned in part 1, we are using trimix 14 / 52 as a diluent.
The percentage of nitrogen in the diluent mix will be 100 – (14 + 52) = 34, that is 34 per cent. And the PN2 of the trimix diluent 14 / 52 at MOD will be 7 x 0.34 = 2.38 bar.
Therefore the PN2 differential on switching from CCR to the bailout gas is 2.80 – 2.38 = 0.42 bar. That’s within the 0.5 bar limit, so we can set the deep bailout gas as trimix 22 /38.
What shallow bailout gas?
 The next question is what to use as our shallow bailout gas and at what depth do we change from the deep bailout? The factors that govern this are:
The next question is what to use as our shallow bailout gas and at what depth do we change from the deep bailout? The factors that govern this are:
- Maximising inert gas off-gassing.
- Minimizing decompression obligation.
- Balancing the cylinder capacities with gas requirements.
- Isobaric counter diffusion.
Clearly, we need to maximise inert gas off-gassing. The largest possible differential between the inert gas partial pressure in the tissues and inert gas partial pressure in our breathing gas will produce this. This must be tempered by the need to remain within our decompression model limits, ensuring we remain within the ICD safe PN2 range.
It would also be handy to balance the cylinder capacity so that we carry similar capacity cylinders for both deep an shallow bailout. It is perfectly possible but maybe a bit unbalanced to carry an 11-litre on one side and a 3-litre on the other; an extreme example but it makes the point.
The strategy then is to change bailout gases at a depth that evens out the gas use as much as we can but allows efficient off-gassing. This tends to mitigate more towards nitrox 50 percent, rather than 80 percent or 100 percent since we will be able to change to 50 percent at a deeper depth.
So assuming nitrox 50 per cent as the shallow bailout gas and wanting a PO2 of 1.6 bar at the change point, the depth we change, calculated using Dalton’s Law again, will be 1.6 / 0.5 = 3.2 bar, that is a switch depth of 22m.
Checking the ICD requirements of this switch as before, we see that the PN2 of nitrogen in nitrox 50 at 22m is 3.2 x 0.5 = 1.6 bar.
And the PN2 in trimix 22 / 38 at 22m is 3.2 x 0.4 = 1.28 bar.
The PN2 differential is 1.60 – 1.28 = 0.32 bar, which is within 0.5 bar limit, so we can use nitrox 50 as our shallow bailout gas.
Additional strategies
We could change to 100 percent oxygen at six metres in order to accelerate the off-gassing, assuming we have open circuit
access to our on-board oxygen. This would aid off-gassing efficiency as well as reducing decompression obligation. However if the bailout was a result of lost oxygen supply, it’s not an option. Alternatively, we could carry a third stage cylinder of oxygen for this purpose or arrange for a staged bailout gas at the decompression station or alternatively as a drop tank as part of team diving protocols.
Another alternative is semi-closed mode operation of the rebreather – an emergency gas extending technique that can be used as long as the breathing loop is intact, functional and with no carbon dioxide problems – but this is a gas conservation strategy that impacts upon bailout gas choice. The diver must have the facility to inject diluent or off-board bailout gas into the breathing loop. This technique relies on using the oxygen content of the diluent and/or bailout gas to partially rebreathe and thus extend the gas supply. For example, if a bailout gas has an oxygen content of 20 percent and we only metabolise 4 percent of the oxygen each breath, you can take a number of breaths from the loop filled with that gas before the oxygen is depleted to a dangerous level. At this point, the diver empties the breathing loop and refills it with fresh gas. The decompression profile is then calculated based upon the lowest fraction of oxygen at each phase of the ascent. It is a complex technique that requires careful planning and good technique in operation.
As a diver ascends, the gas in the loop becomes less dense, lowering the oxygen concentration until a deep bailout gas becomes unable to support the same number of breaths from the loop as it could at depth. At this point, you need to change to a gas with a higher oxygen content, such as your shallow bailout gas. The choice of bailout gases, therefore, becomes critical and on trimix dives the rich nitrox needs to be available at a reasonable depth so that the rebreathe ratio can be maintained while the oxygen content is not reduced to a dangerous level, making 50 percent a good choice.
A deep bailout gas with the highest safe PO2 at target depth combined with a shallow bailout gas with a similar PO2 and that can be used at relatively deep depth is a good compromise. Again if we have access to 80 percent or 100 percent at 6m it will significantly shorten our stay.
The total package
 So the gas choice for a 60m dive, taking into account diluent (discussed in Part 1) and bailout (discussed here in Part 2) gas planning, comes out as:
So the gas choice for a 60m dive, taking into account diluent (discussed in Part 1) and bailout (discussed here in Part 2) gas planning, comes out as:
Diluent trimix 14 / 52
Deep-bailout trimix 22 / 38
Shallow-bailout nitrox 50 percent
Six-metre bailout nitrox 80 percent or oxygen (If available).
One final adjustment we can consider to minimise our open-circuit gas requirement is shortening the ascent phase of the dive by accepting a less conservative ascent profile. Here we balance the risk of decompression illness against the need to carry more bailout gas.
For example, you may choose to reduce the conservativeness when planning a bailout ascent. On some deco models, this may involve simply adjusting a conservativeness level. For example, adjusting down to level 0 rather than 2. On others, the gradient factors may need to be increased such that instead of say 40 / 80 one might choose a less conservative 50 / 90. I merely use these figures as illustrative, not guidance.
Can we standardise?
You might now be asking, do I have to go through this complex gas-planning process every time I do a rebreather mixed-gas dive? The short answer is no.
You will find regular depths and durations that you prefer and it is easy to have a range of pre-prepared gas choices to cover them. With experience you’ll get to know what is appropriate for the dives you do. Modern dive planning programmes do all of the number crunching for you including END, gas density and ICD.
It is important that mixed-gas rebreather divers understand the process and have a good idea of the factors that govern our gas choices and why. You are then able to make informed choices. It is up to each diver to understand the risks and make choices based on their personal risk evaluation and acceptance. This level of diving is not about doing things by rote, in my view. Be adventurous, use the tools to get out and see the sights most others don’t get to see, but do it with knowledge and skill.
See you in the deeps.

 Author: SCUBA | Posted 09 Jan 2018
Author: SCUBA | Posted 09 Jan 2018


